What are Energetic Materials?
Energetic materials are an essential and often unappreciated vital component of all ammunition, without energetic materials an army battalion would be just become a military parade. Energetic materials are used in every step of the munition functioning from the decision to fire until the destruction of the target.Energetic materials are not remarkable in terms of their energy content, 1lb of butter will release more than twice as much energy when burnt than 1 lb. of Trinitrotoluene (TNT) 1 but you can’t demolish a bridge with a stick of butter. Energetic materials’ utility lies in the rate they can release energy, TNT has a “rate of reaction” , detonation velocity, of 6,900 m/s ~ 15,434 mph (think London to New York in 13 minutes)
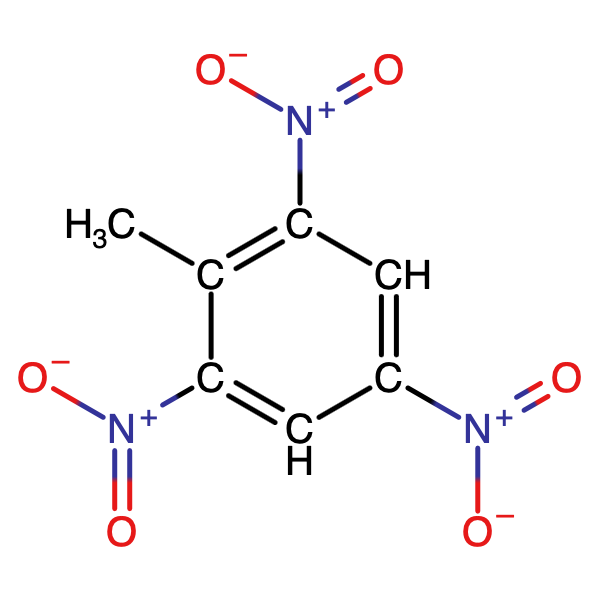
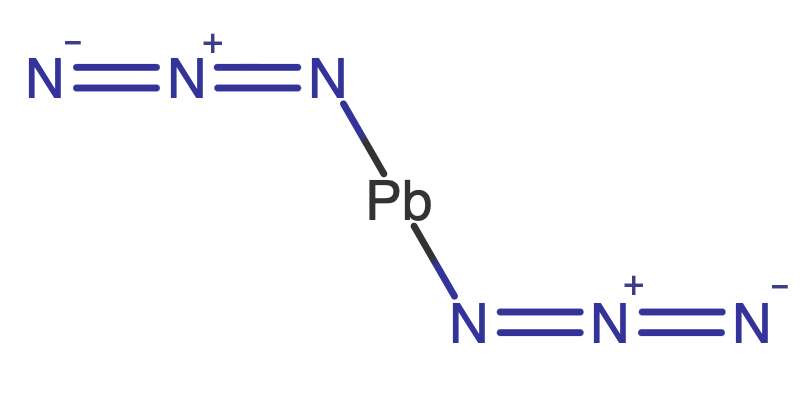
The reason that energetic compounds are able react so quickly is because they do not depend on the availability of atmospheric oxygen, it is either, bound up in their chemical composition or present in other compounds in the formulation. TNT, our all-time favorite energetic material, has most of the oxygen it needs to react right there in its molecular structure close to the carbon and hydrogen atoms which are so desperate to get their hands on some, Figure 1. It doesn’t quite have enough oxygen for complete reaction, see oxygen balance, but it has enough so that it doesn’t have to wait around for the surrounding air to give up its oxygen to start reacting. However, it is not just the proximity of oxidizers that enables this energy to be released, some energetic materials have energy bound up in their structural bonds that is released on decomposition. Some high nitrogen containing materials don’t technically have fuels or oxidizers but nonetheless release considerable energy forming nitrogen e.g. Lead Azide, PbN6 ? Pb + 3N2 , Figure 2.
The functional groups in organic chemistry that give organic compounds explosive properties such as the “nitro” in Trinitrotoluene and the “azide” in Lead Azide are known as Explosophores. Energetics are defined as either high or low explosives depending on whether their burning rates are less than or greater than the speed of sound, see deflagration and detonation A simple classification of energetics is shown in Figure 3.
Pure explosive compounds are listed in Explosive Compounds along with many of their important properties. TNT is one of the few energetic materials that can be used, as is, in artillery, mortar and demolition charges2 without additional ingredients. However, most high explosives are mixtures of explosive compounds, fuels, oxidizers, binders and other additives, these formulations are listed in Explosive Formulations.
1 ?Hc Butter = 30.5 kJ/g compared to ?Hc TNT = 15.0 kJ/g
2 TNT melts at 80.4°C (178°F) which allows it to be melted in water jacketed vessels and then frozen into a solid for operational use.

Explosive Compounds
This table contains over 300 high explosive compounds, some in common use and some new molecules that haven’t made it out of the lab yet (in fact there are some that are mere theoretical possibilities such as N4). The table is initially sorted by CAS number when this is available, as CAS numbers are assigned chronologically this gives a sense of the history of the explosive development. This table is evidently a work in progress and always will be, I am constantly adding to it every time I find a paper on a new compound. My criteria for inclusion in this table is that I can find the chemical structure, and that any additional information is already in the public domain. References in the table are very patchy at the moment but I’m working on correcting this gradually as I find data on density and detonation velocity etc.
Explosive Formulations
Explosives in this database are explosive compositions, that is to say that can be used directly in practical applications (many explosives compounds are too sensitive to use in their pure form these can be found on the Explosives page). These compositions are mixtures made from explosives, binders, plasticizers, oxidizers and fuels.


Pyrotechnics